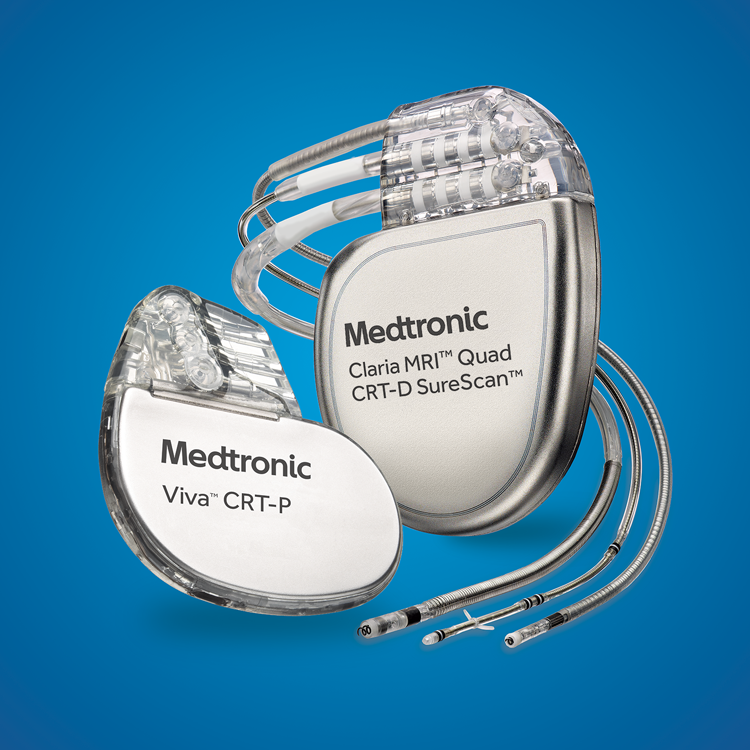
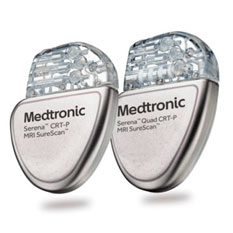
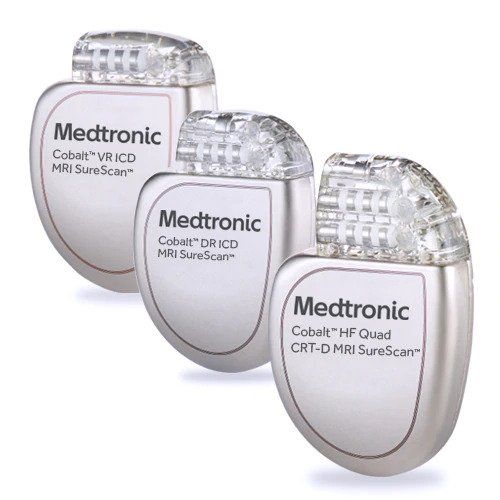
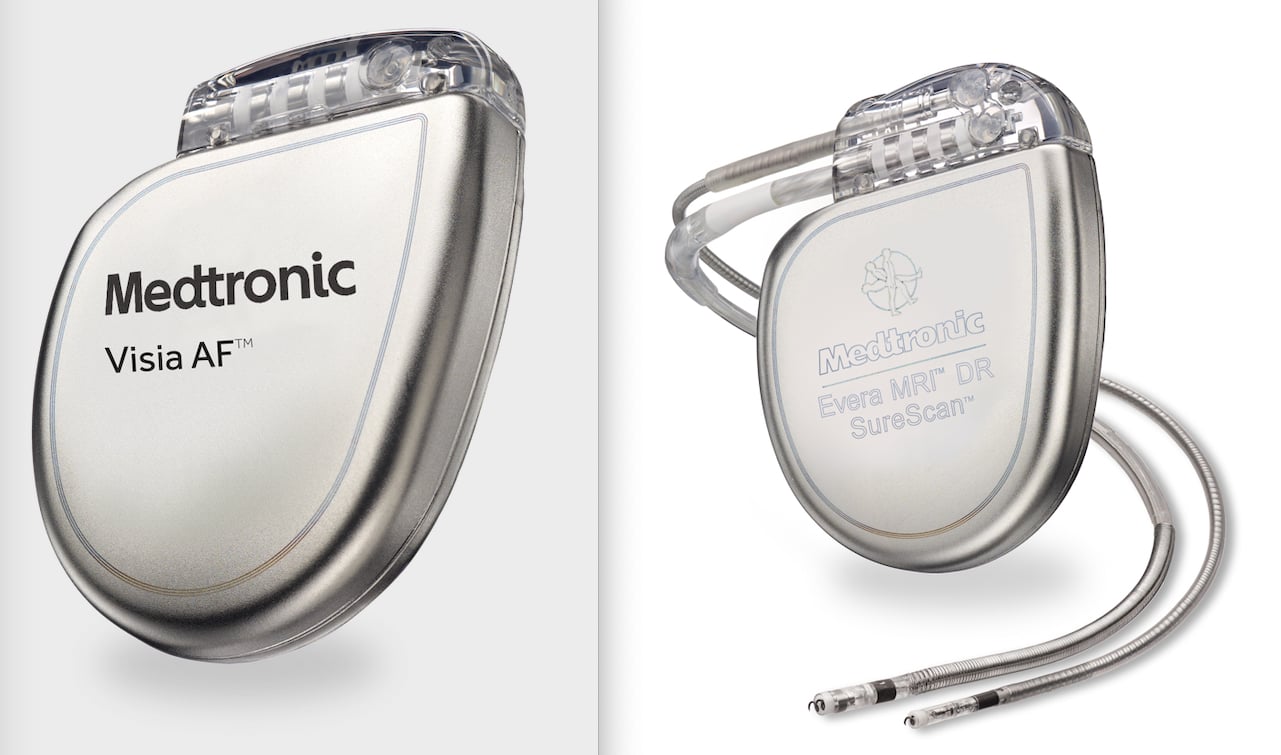
Medtronic India - The Cardiac Resynchronisation Therapy is a clinically proven treatment for certain patients with heart failure. It improves the heart's ability to pump blood and oxygen to the body with

New Analyses Confirm Patients with Medtronic CRT Devices Experience Less Atrial Fibrillation, Are More Active - iData Research
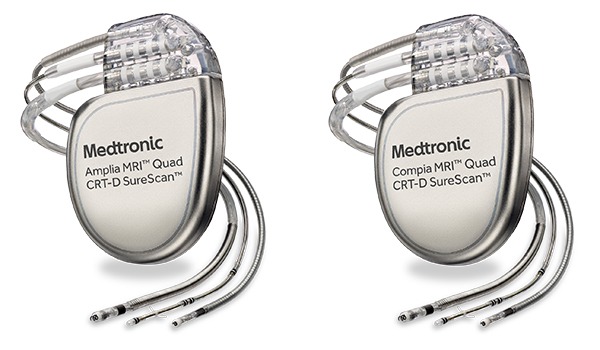
Medtronic receives CE mark for three MRI-compatible cardiac resynchronisation therapy defibrillators - Cardiac Rhythm News